A call to arms
Inside Canada’s impossibly high-stakes rush to lock down tens of millions of doses of the most sought-after product on Earth
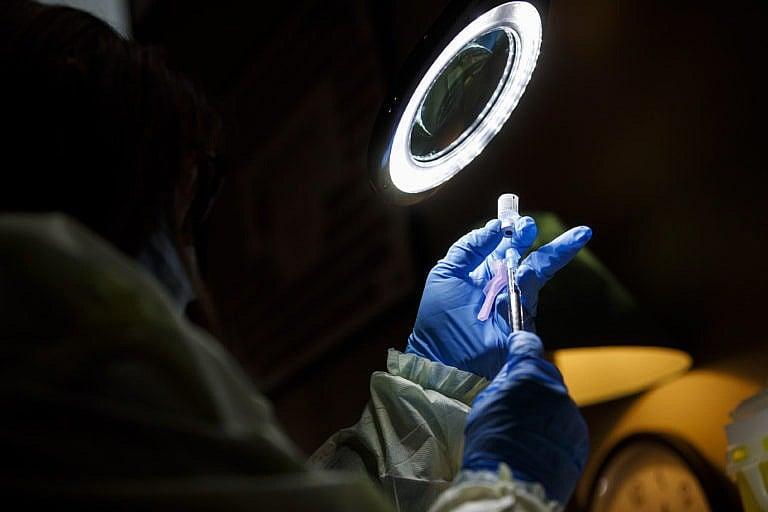
A technician prepares a Pfizer-BioNTech COVID-19 vaccine at a hospital in Toronto (Cole Burston/Bloomberg /Getty Images)
Share
When Agnes Mills was a young teenager, she battled tuberculosis for three years in a hospital in Aklavik, a remote hamlet nestled in the Mackenzie River delta north of the Arctic Circle. She contracted the disease when an epidemic hit the residential school where she lived. She led a solitary existence at the Anglican-run school known as All Saints, separated from her mother, who lived nearby. She was a long way from Old Crow, the tiny Vuntut Gwich’in community in Yukon where she grew up and her grandfather was chief.
Mills, now nearly 85 years old, calmly explains those devastating formative years in Aklavik. She was sexually abused by older girls at the school. An extended hospital stay offered a break from All Saints, but Mills says she received little education.
Eventually, Mills moved south. She lived in Edmonton and Kenora, Ont., before settling in Ottawa where she was the executive director of the Native Women’s Association of Canada. She worked in government for a decade before returning north, where she was an Elder adviser of the Truth and Reconciliation Commission. Last year, Mills received the Order of Yukon.
Now she lives at Whistle Bend Continuing Care Facility in Whitehorse. Dementia is creeping into her life, and most of a year without seeing her grandchildren has been excruciating. “It’s a very lonely time. You’re all by yourself in this little room,” she says.
On Jan. 4, Yukon started immunizing high-risk residents with Moderna’s COVID-19 vaccine. Mills, a survivor of so much hardship, was at the front of the line.
About 50 residents and staff at the Whistle Bend home got their first shots that day. The 150-bed facility has managed to avoid an outbreak since last March—a feat matched by every other home in Yukon, where only 70 cases have been reported across the entire territory.
Dozens of long-term care facilities across Canada haven’t been nearly as fortunate. Of the roughly 20,000 Canadians who succumbed to COVID-19 in the pandemic’s first 11 months, nearly 9,000 were residents of long-term care homes in Ontario and Quebec—a grim number that climbs daily.

As new, more easily transmissible variants of the disease threaten to spread quickly among vulnerable communities in Canada, the need for widespread vaccinations has never been more urgent.
Mills’ single inoculation was an early milestone in Canada’s desperate, stubborn quest for normalcy—a remarkable but still infinitesimally small step in a massive immunization campaign, the largest in Canadian history, meant to beat COVID-19.
That campaign needs doses, and a federal bureaucracy upended by the pandemic has been duking it out with the richest countries in the world for what is now the hottest commodity on the planet. Without the ability to produce COVID-19 vaccines at home, Ottawa is bargaining with a powerful pharmaceutical industry that holds all of the cards. And the feds are racing against the clock. Every day without vaccines means more sickness, tragedy and preventable death.
***

Bergstedt moved to Moderna after nearly 20 years at Merck (Photograph by Kayana Szymczak)
June 1 was Patrick Bergstedt’s first day on the job as Moderna’s senior vice-president of commercial vaccines. Bergstedt is an affable South African, who was based in the small town of New Hope, Penn., at the time. Like so many of us, his office was in his home, and he was making deals over Webex.
“I was commercial person number one,” he tells Maclean’s, describing the emerging biotech player as a “tiny little company that had never sold anything in its life.” He wasn’t exaggerating. Moderna was developing a wide variety of products, but had yet to make a sale.
Bergstedt came from Merck, a pharmaceutical giant where he spent nearly 20 years and gained familiarity with Canada’s health system, which he viewed as “very progressive” on vaccinations. “You can have vaccines sitting in a warehouse, but it’s about vaccines in arms,” he says. “That’s what’s important.”
For all his years in the business, Bergstedt had no idea who to call north of the border about COVID vaccines. On June 11, as global interest in Moderna’s mRNA vaccine was starting to pick up, a serendipitous email landed in Bergstedt’s inbox from a couple of Canadians.
Bergstedt might have expected to hear from a scientist or procurement officer, but it was friendly neighbourhood diplomats who came calling. Valérie La Traverse and Carolin McCaffrey, a senior trade commissioner and an investment officer at Canada’s consulate in Boston, reached out to learn about the fledgling company’s vaccine and gauge interest in potential expansion to Canada.
“There is a God,” thought Bergstedt.
In Ottawa, it was all hands on deck—even Global Affairs Canada had been pressed into duty to work contacts that might kick-start vaccine talks or dredge up some leads.
Within a week, Bergstedt was on a call with then-consul general David Alward. On June 26, he was back on a video conference with representatives of Health Canada; the Public Health Agency; Innovation, Science and Economic Development; and Public Services and Procurement. They peppered him with questions about clinical data and potential supply. “I’ve been on many calls with many different countries,” he says. “But I have never had a call that big, representing so many branches of government.” Bergstedt recalls that Moderna’s reps all had their webcams turned on. The federal officials, he notes, kept theirs off.
In these early conversations, Moderna’s vaccine was promising, but unproven. The company had created the vaccine in 42 days and dosed the first test subject three weeks later.
“At that time, we had no clinical data,” says Bergstedt. “We had this early, experimental phase 1 data that says this vaccine produces an immune response. We had no data on efficacy or safety.”
Even to Bergstedt, the eager salesman, getting the vaccines into arms before the end of the year seemed an impossible task.
***
Within days of Moderna connecting with Ottawa, Mark Lievonen was sitting in his condo in Stouffville, Ont., when he pulled out his iPad and logged onto Zoom. For many years, Lievonen had been the president of the Canadian vaccine division of Sanofi, a pharmaceutical giant. Now he joined Joanne Langley, a Dalhousie University professor and head of infectious diseases at Halifax’s IWK Health Centre, as co-chair of the federal COVID-19 Vaccine Task Force (VTF).
As the pandemic’s first wave was slowly subsiding in hard-hit Ontario and Quebec at the end of May, Lievonen and Langley were invited to head up the group. They were asked, says Langley, to serve their country. “When you get a letter like that, you just say yes.”
The federal government assembled an initial group of 12 core task force members, including epidemiologists, physicians, manufacturing specialists and industry insiders, with orders to recommend safe and effective vaccines the feds should buy in large quantities—a daunting assignment, since none of the leading candidates were anywhere near a sure thing. “As we were about to begin, I thought, this is a mind-boggling, important task,” says Lievonen. “What if we fail? That was in the back of my mind.”
A few weeks before the task force got to work, Lievonen had made a presentation—by Zoom, of course—to a public COVID-19 webinar at the University of Toronto’s Dalla Lana School of Public Health. He cautioned his audience that a novel coronavirus vaccine could take well over a decade to produce. Some observers had pegged 12 to 18 months as the fastest possible time frame, which Lievonen recalls was “still far beyond anything I would have thought at that point.”
Starting in June, the task force quietly invited manufacturers of most of the leading candidates to make presentations. (The panel’s existence would not be made public until August—and even now the government is reluctant to talk about it.)
“Most members were logging full-time hours, particularly through July and August,” says Lievonen. They liked what they heard from Moderna, Pfizer-BioNTech, AstraZeneca, Johnson & Johnson, Novavax, Medicago, Sanofi and GlaxoSmithKline—and eventually recommended that Public Services and Procurement Canada, headed up by rookie cabinet minister Anita Anand, pursue supply agreements with all of them.
While those experts toiled in obscurity, the government’s critics were already beginning to raise the alarm about a weak link in the federal procurement strategy. Other countries, like the U.K. and the U.S., had plans to manufacture vaccines by the millions on their own soil, but Canada did not; nor could it rely on American supply chains, which Washington had reserved for domestic supply.
Canadians, it turned out, would be entirely at the mercy of European-made drugs.
***

Prime Minister Justin Trudeau speaks with scientist Krishnaraj Tiwari, left, as Minister of Economic Development Melanie Joly, Minister of Science and Industry Navdeep Bains and NRC President Iain Stewart look on during a visit to the National Research Council of Canada's Royalmount Human Health Therapeutics Research Centre facility in Montreal on Aug. 31, 2020. (Graham Hughes/CP)
By early August, Brad Sorenson had been waiting to hear back from Ottawa for several months. Before the World Health Organization declared a global pandemic in March, the president of Calgary-based Providence Therapeutics had started talks with the feds on a homegrown COVID-19 vaccine. But he was starting to feel impatient.
As far back as February, Sorenson conferred with his scientific advisory board about the novel coronavirus. It was clear to them life was about to be disrupted in a big way. The company had been working on cancer vaccines but decided to pivot to COVID-19.
Sorenson spent part of March in Hawaii for his father’s 80th birthday. He returned to Alberta just before lockdown, and convened an emergency meeting on the development of a COVID-19 vaccine that used mRNA—the same technology as Pfizer and Moderna’s product. Within weeks, Providence took a preliminary plan to Health Canada. The bureaucrats were “responsive” and eager to learn more. At that point, Sorenson estimates his company trailed Moderna’s progress by only a couple of months.
By April, Providence had submitted a formal proposal to the federal Strategic Investment Fund that sought $35 million for clinical trials. Then the waiting began. Days turned into weeks, and weeks turned into months. On Aug. 17, the day before the Liberals prorogued Parliament, Sorenson heard back. The feds had turned down the proposal, saying Providence wasn’t far enough along to meet the requirement for funding. Eventually, the company scored a few million dollars for clinical trials from the National Research Council (NRC). Sorenson admits he was angry at the time. “This was no warp speed,” he says. “This is turtle speed.”
As Sorenson waited, his proposal started to look like a missed opportunity for a federal government desperate for domestic vaccine production.
China’s CanSino was Canada’s big early bet on making a vaccine domestically. Attached to the plan was $44 million for upgrades to the NRC’s Human Health Therapeutics facility in Montreal. That plant was meant to eventually produce CanSino’s vaccine candidate, Ad5-nCoV.
The plan fell through when Chinese customs officials prevented the shipment of test vaccines from leaving the country. Documents tabled in Parliament in January revealed the feds were aware of the blocked shipment just days after the CanSino deal was announced in May. Exactly why the shipment was blocked remains a mystery, though the Chinese didn’t kibosh similar exports to other countries. Canada’s bet on CanSino angered the government’s critics, including the opposition Conservatives, who cast the Liberals as naive for partnering with a Chinese firm in the middle of a deep freeze in bilateral relations.
But if not CanSino, could the NRC lab be used to produce another company’s more promising candidate? Other countries had plans to manufacture AstraZeneca’s vaccine on their own soil. Why, critics wondered, hadn’t Canada negotiated a similar licensing agreement?
The Liberals insisted that was impossible at the time. A spokesman for Public Services and Procurement Canada says when the feds were doing deals, the country “had no flexible large-scale bio-manufacturing capacity suitable for a COVID-19 vaccine.” Canada hasn’t had a publicly funded lab that mass-produces vaccines since the ’80s, and no big private labs have the technology in place to make mRNA vaccines. Both of the largest pharma facilities in Canada—GlaxoSmithKline’s plant in Quebec City and Sanofi’s operation in Toronto—would have had to build entirely new facilities in order to produce a vaccine like AstraZeneca’s, says Andrew Casey, the president of pharmaceutical lobby group BIOTECanada.
In August, Ottawa committed $126 million over two years for a new NRC bio-manufacturing facility in Montreal. It said at the time the new facility would, as early as this coming July, be equipped to produce up to two million doses a month of several kinds of COVID-19 vaccines—including the viral vector technology used by AstraZeneca and the protein subunit technology used by Novavax. (That projected date would prove to be wildly optimistic.)
The feds could also have given Providence a shot. Sorenson laments what could have been with that $35-million injection last year. Providence, which is currently running phase 1 clinical trials, might have been much further down the line.
CanSino, meanwhile, found a new locale to test its vaccine: Russia.
***

Anand, Minister of Public Services and Procurement Canada, oversees the off loading of COVID-19 vaccines on Dec. 13, 2020 (Courtesy of Cpl Matthew Tower/DND)
Canada’s hope for nailing down contracts for vaccines now rested with a tiny group of bureaucrats, just 10 to 20 people working from home in and around Ottawa—and Oakville, Ont., where Procurement Minister Anita Anand spent the summer.
“You have to keep it small because we put our best negotiators on the table,” explains Arianne Reza, the associate deputy minister for procurement at Public Services and Procurement Canada.
They were a seasoned team already battle-tested by months of pandemic scrambling, says Reza. The department was just emerging from the springtime rush for personal protective equipment and testing supplies. They were buying billions of products seemingly on the fly. “You normally do business with pre-qualified, trusted suppliers. All of a sudden, we were in the thick of it, buying masks and gloves,” says Reza. “So much complexity. So much risk.”
At one point, when Reza turned to procuring all the supplies for immunization—syringes, vials, stoppers and swabs—she picked up the phone and called a vendor out of the blue. “That person was very suspicious of getting cold-called by the Government of Canada to offer them a contract,” she says. “I had to go through many layers, many verifications, to convince them I was the real deal.”
The feds bought up to two billion pieces of PPE, 40 million rapid tests, 145 million syringes and 126 cold and ultra-cold storage freezers. Even so, the negotiations for vaccines was a whole new game.
Anand, a lawyer and professor who specialized in corporate governance before entering politics, explained the intricacies of vaccine deal-making. “It’s not simply a matter of distributing standard-form contracts and having vaccine suppliers agree to our preferred terms,” the procurement minister tells Maclean’s. “Each supplier has its own set of concerns. As a result, each agreement is bespoke and contains terms related to doses and price and manufacturing and finishing parameters of each vaccine.” The manufacturers, keen to see doses actually administered, were lured in part by Canada’s strong public health network and widespread embrace of vaccination.
Reza said Canada enjoyed certain advantages. “The reputation of having a first-rank regulator made it easy to attract them to the table,” she says. A senior government source familiar with vaccine procurement planning says Canada faced a “middle power issue”—wielding more influence than most countries, but offering smaller orders than the U.S. or European Union. Reza took the opposite view, arguing that Canada’s comparatively modest needs “wouldn’t add a huge burden” to manufacturers’ supply chains.
In the case of Moderna, Bergstedt says negotiations involved “tough conversations” that stretched late into the night and into weekends as the two sides haggled over the timing of supply and the volume of doses. The Canadians were “efficient,” he says; it had been more of a challenge to pin down negotiations with EU customers. Moderna knew the whole planet, including Canadians, were watching closely. “Building a relationship of transparency and trust is critical for us in a world in which people are very cynical about pharma in general,” says Bergstedt.
On July 24, Canada inked an advance supply agreement with Moderna. The feds announced other deals: Pfizer on Aug. 5, Johnson & Johnson and Novavax on Aug. 31, GlaxoSmithKline on Sept. 22, AstraZeneca on Sept. 25 and Medicago on Oct. 23.
By early fall, Canada seemed to be sitting pretty, having secured up to 398 million potential vaccine doses between seven companies, the world’s most diverse portfolio and more doses per capita than any other nation on Earth.
Some questioned Ottawa’s refusal to discuss the fine print of those deals—what was Canada paying and what did the contracts say about Canada’s place in the global pecking order? Canada was one of the first countries to lock down deals with Moderna and Pfizer, but was much later than some of its rivals to sign with other companies. Could that affect deliveries?
“Staking out the back of the queue means that we might beg for a little vaccine in a hurry for our health-care workers or most vulnerable,” Amir Attaran, a biomedical scientist and lawyer at the University of Ottawa, wrote in Maclean’s last summer. But ordinary Canadians should “expect to wait.”
Procurement Canada’s official line was the feds “cannot disclose details of specific agreements in order to protect Canada’s negotiating position and commercially sensitive pricing information, as well as to respect confidentiality clauses in the vaccine agreements made to date.”
Nevertheless, early doses would soon begin arriving, and months ahead of anyone’s wildest expectations.
***

Arnold Roberts, 93, was the first Ottawa care-home resident to get vaccinated (Courtesy of Darren Brown/Ottawa Public Health)
Four days after Moderna’s vaccine was approved on Dec. 23, a FedEx delivery arrived in Whitehorse. Similar deliveries would happen over Christmas in all three territories.
Benton Foster, Yukon’s director of community health programs, said his team “first started hearing rumblings” about the distribution of the Pfizer and Moderna vaccines in early November, before either was approved. The feds were hinting at first-quarter deliveries in 2021, which Yukon officials initially interpreted as aligning with their fiscal year—meaning they’d see delivery in April. They soon realized Ottawa was talking about the calendar year. “Everyone was kind of in disbelief that it would happen,” says Foster.
The rollout required a big country like Canada, with so many rural and remote communities, to reckon with monumental logistical questions. Former military leaders, known for their mastery of rules and promptness, emerged as obvious choices to lead the effort. The feds enlisted Maj.-Gen. Dany Fortin to coordinate federal-provincial co-operation. Ontario turned to Rick Hillier, Canada’s chief of the defence staff during the height of the war in Afghanistan. UPS and FedEx unveiled advanced tracking technology that monitored every shipment from Europe.

A vaccination kit at a Montreal clinic (Paul Chiasson/CP)

A UPS employee delivers a box of Pfizer-BioNTech vaccines in Montreal on Dec. 14 (Andrej Ivanov/AFP/Getty Images)
But a less hopeful Christmas story was unfolding in Ontario, where the real challenge with the vaccine rollout was proving to have little to do with logistics.
On Boxing Day, health-care workers in Ontario took to social media in protest after the province stopped vaccinations for a short period over the holidays. Observers also noted a significant drop in injections on weekends. At a time when more than 100 people in Ontario were dying every day in an out-of-control second wave, a pause in vaccinations looked as absurd as firefighters leaving a five-alarm blaze for a lunch break.
A source close to Ontario’s rollout team stood by the province’s strategy, arguing that health-care workers desperately needed a holiday break, and the province was ratcheting down weekend appointments largely to prevent no-shows that would result in thawed doses going to waste.
But a bigger problem still was emerging across the country. Most provinces’ hastily prepared immunization programs started to outpace shipments from Pfizer and Moderna by January. Several provinces worried they’d run out of doses before they could administer second shots within the roughly three-week time frames recommended by Pfizer and Moderna.
The Liberals were nevertheless sticking to their guns. Negotiators had secured four million doses of the Pfizer vaccine and two million doses of Moderna’s product, guaranteed before the end of March. And Canada would still get those doses, they promised.
The truth was that the numbers were now out of the government’s control.
In mid-January, Pfizer announced a temporary slowdown in production as the company retooled its facility in Puurs, Belgium. Christina Antoniou, a company spokeswoman, acknowledged at the time that “fluctuations in orders and shipping schedules” would impact deliveries “in the immediate future.”
A few days later, the Liberals dropped a bomb. Pfizer would send no shipments to Canada in the last week of January. Federal officials claimed every country would receive fewer doses, and that they expected “equity” from Pfizer. But exports to EU members never hit zero.
Israeli Prime Minister Benjamin Netanyahu, whose country emerged as a global vaccination leader, boasted of a dozen phone calls with Pfizer CEO Albert Bourla. Suddenly, everybody was calling Pfizer Canada’s president, Cole Pinnow. Doug Ford called him up. Erin O’Toole picked up the phone and then demanded Trudeau call Bourla—which he did.
Anand maintains “very close contact” with Pinnow. “I text with him regularly,” she says, adding that she spoke to him every day the week Pfizer announced reductions. Moderna gets the same treatment. Patricia Gauthier, the company’s country manager for Canada, says phone calls certainly don’t hurt. “Everybody’s looking at how can we accelerate? What can we do? I understand people are anxious.”
On Jan. 29, Moderna also announced a temporary reduction in shipments to Canada.
Amid the uncertainty, NDP MP Don Davies, the party’s health critic, joined the Tories in pressuring the Liberals to release at least some of the terms and conditions of their contracts, as the U.S. had already done. “I’m very suspicious of a government that’s gone through such Herculean efforts to resist that. What are they hiding?” he asks. “Deliveries are probably contingent on a lot of factors that are out of the government’s control.”
Moderna’s Bergstedt offers that the company will face penalties if it misses its first-quarter obligations. “The government has held the industry responsible for keeping its commitments,” he said. “We’ve not been given a blank cheque.”
In the middle of the zero-delivery week, Canada’s place in the queue took yet another potentially disastrous hit when the EU, angry with U.K.-based AstraZeneca for reducing its planned supply, threatened export controls. Trudeau told reporters that European Commission president Ursula von der Leyen had assured him EU protectionism wouldn’t affect Canadian imports. But Canada was absent from a list of exempt countries.
The limited supply of approved vaccines meant that one country’s gain was necessarily another’s loss. That contagious vaccine nationalism threatened to launch a supply chain cold war.
At the end of the month, Canada’s sputtering vaccine effort ranked 20th in the world, behind countries like Spain, Hungary and Estonia.
***

Paramedics prepare COVID-19 vaccines at the Extendicare West End Villa in Ottawa (Courtesy of City of Ottawa)

Health-care workers at a Toronto vaccine clinic (Nathan Denette/CP)
In early February, Canada announced a new plan to manufacture Novavax doses in Montreal, beginning in 2022—months after the initial vaccination campaign is supposed to wrap up at the end of September. In the likely event new variants require new or modified vaccines, Canada will be ready.
But Canadians will surely be looking enviously at countries that will ramp up much sooner. Australia will be pumping out AstraZeneca doses by the millions domestically as soon as that shot is approved. Japan plans to manufacture up to 90 million doses of the same vaccine.
Domestic production and licensing agreements increasingly seem to be the path to speedy vaccinations. The U.S. is ramping up quickly, having administered 32 million vaccines by February, three times Canada’s per capita rate. At the same time, the U.K. was at five times Canada’s rate.
Israel bet on closer ties with Pfizer, agreeing to hand over anonymized biometric data in exchange for early doses—a deal that raises serious privacy concerns but has also seen five million shots administered in a month. It’s already seeing sickness declining among those over 60.
Canada’s solution in the absence of an ability to manufacture vaccines was an impressive shopping spree—we out-purchased everyone in the world. In February, along with AstraZeneca’s imminent approval, both Novavax and Johnson & Johnson published clinical data that showed promise. Canada had secured up to 134 million doses of those three vaccines. The single-dose Johnson & Johnson vaccine could on its own be a game-changer.
Still, at this writing at the beginning of February, the hole in Canada’s plan has been deepening by the day, as the rollout falls further and further behind other developed countries. Can Canada catch up? Maybe. If supply disruptions don’t sink the government’s planned ramp-up in April, Canada would need to administer more than 300,000 shots per day before the end of September to inoculate most Canadians. That’s orders of magnitude more than the current peak this year of 48,195 on Jan. 15. But, again, if the deliveries that Canada’s devoted team of bureaucrats lined up last spring start landing as planned, the pieces are in place to get doses into arms in community halls, arenas and pharmacies across the nation.
The priority for now remains long-term care homes, where new, more transmissible COVID variants have started to creep in. More than 50 people died at a long-term care home in Barrie, Ont., in January. The province planned to immunize every resident and worker in high-risk long-term care homes by Feb. 15.
Ontario hopes to extend its rollout to other high-risk populations—those over 80 years old, front-line workers and Ontarians with high-risk conditions—in March. In August, it will open up to anyone who wants a shot. British Columbia is hoping to broaden beyond high-risk populations by July. In a “high-supply scenario,” Manitoba plans to wrap up its program entirely by September. Alberta, on the other hand, only anticipates a rollout to the general public in the fall. A general rule of thumb: the youngest will wait the longest.
But there’s no real winning in this race. Canada has lost 20,000 souls and counting. There isn’t a vaccine on the planet that can fix that.
This article appears in print in the March 2021 issue of Maclean’s magazine with the headline, “A call to arms.” Subscribe to the monthly print magazine here.
