Building a better human
Scientists are already able to replace many worn-out or missing body parts with new ones. But two-thirds of a person?
James Cheadle / Smithsonian Magazine
Share
Growing a human organ is a bit like baking a layer cake, says Dr. Anthony Atala, director of the Wake Forest Institute for Regenerative Medicine. Let’s say the “cake” we want is a kidney. After harvesting cells from the patient’s kidney and coaxing them to multiply—mixing up the cake batter—Atala’s team bastes those cells onto a biodegradable scaffold, one painstaking layer at a time. “Once there’s the right amount,” he says, “we put it in an oven-like device that has the same conditions as the human body.” The kidney “bakes” inside the bioreactor for a couple of weeks, where it’s also exercised. Then it’s ready for implant. Eventually, the scaffold melts away, leaving the new organ.
A donor kidney was the first organ to be successfully transplanted into a patient, in 1954. Five decades later, we’re building new ones from scratch—growing them on scaffolds or printing them with modified desktop printers that shoot cells instead of ink. About 14 years ago, Atala’s team implanted bioengineered bladders into patients and, he says, “they’ve lasted all these years.” He and other scientists are moulding jumbles of cells into heart valves, ears, stomachs and skin. They’re building advanced prosthetics, including bionic hands and legs, which mimic natural function and can even be controlled by our minds. More and more people will live their lives with these artificial parts integrated into their bodies.
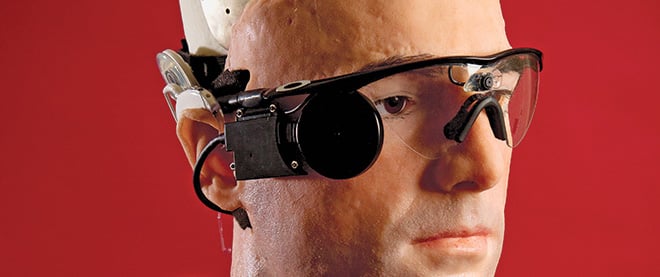
The pace of discovery has moved so quickly, it’s caught even some experts off guard. A team of scientists, roboticists and engineers recently got together to see how much of a human being they could create from the ground up—a Bionic Man, using lab-made parts that are already used in people, or will be soon. “I was surprised by how much [of a person] we were able to build,” says roboticist Rich Walker, managing director of Shadow Robot Company, who oversaw the project. The Bionic Man, which cost $1 million to make—not to mention the billions of dollars in research it represents—is about 70 per cent complete. It has synthetic organs, including lungs, a pancreas, kidney, spleen and trachea; bionic hands, called the i-limb; synthetic blood pumping through an artificial heart that squeaks as it beats; and, most disconcertingly, a human face mounted on its cyborg frame. Walker mostly builds robots, but the Bionic Man is not a robot. It’s a stand-in for a human—one that, within our lifetimes, some of us will begin to more closely resemble. “We took things that are add-ons to a person,” Walker says, “and put them all together.”
Its face is a replica of Bertolt Meyer’s, a professor at the University of Zurich, who wears an i-limb. Meyer was born missing his lower left arm. Like other electronic prosthetics, the i-limb (from Scotland-based Touch Bionics) has electrodes that press against his residual arm, reading signals given off by its muscles. Its articulated digits improve on other prosthetics’ clunky pinch. Individually powered fingers detect shapes of objects, allowing Meyer to hold a carrot and slice it, carry a bag, or—for the first time in his life—extend his fingers and clap his hands. In the latest version of the i-limb, users program favourite grips into a smartphone and transmit them wirelessly to the prosthetic via Bluetooth. It comes in flesh tones, but Meyer opted for a clear casing so the mechanics within are visible. He likes to show it off. “It comes across as high-tech, like having the newest iPhone.”
Since 2009, when he was fitted with his first i-limb, Meyer’s been an unofficial ambassador for Touch Bionics. Outspoken, genial, and with a social scientist’s perspective (he teaches social and business psychology), he was asked to work on the Bionic Man. “I said yes after I said no twice,” says Meyer, who also hosts a documentary about the project. (The Incredible Bionic Man, a co-production from Channel 4 and the Smithsonian Channel, will air in Canada on Nov. 13.) Meyer raises a slew of disturbing questions, not least of which is who gets to benefit from these technological advances: his i-limb cost over $50,000, but the prosthetic can cost up to twice as much, depending on the needs of the patient. When artificial body parts begin to exceed the abilities of natural ones, “should we allow people to chop off their limbs to replace them with bionic ones?” Once they surpass natural counterparts, he says, “market forces will take over, and there’s money to be made.” Just as we now lust after the latest iPhone, Meyer believes we will soon covet bionic eyes, ears and limbs.
The dream of building a person from artificial parts has always been tempered with uneasiness, a fear of the “unnatural,” of losing sight of what makes us human. (One need look no further than Mary Shelley’s Frankenstein.) Meyer’s first encounter with his bionic doppelgänger provides one of the most unsettling moments of the documentary. As roboticists fix the eerily lifelike silicon face onto its skull, he begins to flush. Standing face to face with a cyborg version of himself, its robotic limbs outstretched, artificial organs exposed, Meyer is visibly uncomfortable. “It looked like my own death mask,” Meyer tells Maclean’s. After briefly staring down this cyborg version of himself, Meyer had to excuse himself and leave the room.
The Bionic Man’s windpipe, arteries, urethra and bladder come from the lab of Dr. Alex Seifalian, a professor of nanotechnology and regenerative medicine at University College London. Other examples of all these lab-grown parts have also been implanted into patients, or are about to be. (The bladder and urethra are undergoing preclinical assessment.) “Right now, we’re making a lot of ears,” he says.
Seifalian, who speaks with a soft Iranian accent, is a pioneer in his field: in 2011, he built a windpipe that was transplanted into a man living in Iceland, the first time that a fully synthetic organ had ever been used for transplant. (Seifalian saw this man in January, almost two years after the operation, and he was “doing very well,” he says.) Recently, Seifalian had a patient who’d lost his nose to skin cancer. In a groundbreaking treatment—the first of its kind—Seifalian produced a nose for the 56-year-old British businessman. But, in a twist that even science-fiction writers might have found too outlandish to be believable, Seifalian grew the nose inside the patient’s own arm.
Seifalian started by making a mould based on the man’s original nose, then created a scaffold from a nanocomposite material. After stem cells from the patient were added to the scaffold (stem cells are the body’s building blocks, and can become virtually any cell type), the nose was put in a bioreactor, where it was left to mature.
Meanwhile, a balloon was placed inside the patient’s arm to stretch out a pocket of skin. Inserting the nose into the arm was necessary, Seifalian says, so that it would acquire a network of blood vessels and its own covering of skin. The whole process took three months; it was transplanted to his face earlier this year. The British man can’t yet smell out of his new nose; within the next few months, Seifalian plans to open up the nostrils and seed them with more stem cells, which should return his sense of smell.
Another method of producing parts, one used by Atala and others, involves taking donor organs and rinsing them with detergents to clear away all the donor cells, then basting them in the patient’s stem cells. “The architecture is really perfect,” Atala says, “because it comes from a real organ. And there’s no danger of rejection, because the donor cells are washed away.” (At the McEwen Centre for Regenerative Medicine in Toronto, a team of scientists is working on producing tracheae for transplant this way.) The downside is that a donor organ is required. Regenerative medicine aims to bypass long-standing challenges of organ donation, like the lack of viable donors and the risk of rejection.
Bioprinters will speed up the process, as printing off a body part is much faster than crafting one by hand. Burn victims should be among the first to benefit from the technology. Today, they’re treated with skin grafts harvested from their own bodies, which “creates a new wound,” says burn surgeon Dr. Marc Jeschke, director of the Ross Tilley Burn Centre at Sunnybrook Health Sciences Centre in Toronto. Growing fresh skin would be ideal, but current techniques take too much time to make it practical. “If you take six to eight weeks to make a sheet of skin, the patient will be almost healed or dead.”
Jeschke is part of a Toronto team developing a bioprinter that will print human skin. Just as an inkjet printer shoots cyan or magenta dots onto paper, the bioprinter dispenses different types of cells, grown from the patient’s own, into a hydrogel matrix. Unlike a desktop printer, it can shoot cells in three dimensions, and has a much faster writing speed. Jeschke’s team is honing the technique with mice, and hope to use it to treat patients in a few years. (The Bionic Man doesn’t have lab-made skin. “We looked at it. It would have been really interesting,” says Walker, the roboticist, “but it would have rotted. It needs to be supported by the body to live.”)
Despite the pace of research, we’re still years away from mass-producing human parts. Some of Seifalian’s work, including lab-grown arteries, have reached clinical trials; others, like the nose and a tear duct conduit, were “compassionate cases” for patients who had no other options. They’ve yet to be approved on a wider basis. “The average for a drug to be approved by the [U.S. Food and Drug Administration], before it reaches the patient, is 15 years,” Atala says. “That’s just for a pill. These will take longer.”
Still, clinical trials of stem cell treatments are popping up. At the Ottawa Hospital Research Institute, a new one aims to fix damaged heart muscle using genetically enhanced stem cells. After a patient suffers a major heart attack, stem cells are extracted from their blood and boosted with a gene that promotes blood vessel growth and tissue healing. Then they’re infused into the heart using a procedure similar to an angioplasty, with the goal of repairing the muscle tissue.
To Atala, human organs fall into one of four categories, ranging from simplest to make to most difficult. First come flat parts, like skin. Then there are tubular organs, including the windpipe and blood vessels. Next are hollow, non-tubular parts, such as the stomach or bladder. The last, and most difficult to create, are solid organs, like the heart, liver, lung and kidneys. “Up to this point, we’ve implanted the first three types,” says Atala. “We have not yet implanted a solid organ.” But it can’t be all that far off. In his lab, he’s growing human fingers.
While scientists sprout body parts in bioreactors, others are mimicking them with machines. Bill Johns, a chemical engineer who lives in Oxfordshire, England, designed an artificial lung after losing his son Graham to cystic fibrosis. “Apart from his lungs, he was fit,” Johns says. “I thought, it’s not brain surgery to put oxygen into blood and take carbon dioxide out.” A prototype of his lung—a small, curved white box with thin pipes that carry blood and air through its chamber—is inside the Bionic Man’s chest. Developed by Haemair Ltd. (of which Johns is managing director), the lung should be available for use outside of the body by 2015. A couple of years later, it may be possible to implant it. “Surgeons are very keen,” he says. In the U.K., only one per cent of patients who need a lung transplant now receive one, because of the acute shortage of lungs.
The Bionic Man is a showpiece for such parts. It “sees” with an electronic retina designed at the University of Oxford—a tiny microchip that has already been implanted into the eyes of patients suffering from retinitis pigmentosa, an incurable condition that leads to blindness. It moves with a robotic exoskeleton designed to help wheelchair users stand and walk. Its “brain” is a chat-bot program that allows it to answer basic questions. It has an artificial kidney and a “spleen-on-a-chip” to filter its blood. A battery-powered heart made by SynCardia Systems, Inc., which is used by patients awaiting transplants, pumps synthetic blood through its veins. The blood’s plastic molecules have an iron atom at their core, like hemoglobin; they bind oxygen in the lungs and carry it through the body. This plastic blood will be ideal in emergency situations. It has a longer shelf life than donor blood and doesn’t spoil if left unrefrigerated.
The next step is to create prosthetics that integrate with our minds as seamlessly as a flesh-and-blood limb. In 2012, a team of U.S. researchers announced they’d made a brain-machine interface that allowed Cathy Hutchinson, who’d been paralyzed for 15 years, to pick up her coffee and take a sip using a robotic arm controlled by her thoughts. In that case, a sensor was implanted in Hutchinson’s brain, but in the future, that shouldn’t be necessary. Another team of U.S. scientists has created a thought-controlled bionic leg worn by Zac Vawter, 32, who lost his lower right leg in a motorcycle accident. Surgeons rewired Vawter’s leg, directing nerves from damaged muscle to the healthy hamstring above his knee. When nerves told the muscles to contract, their signals were picked up by sensors in the prosthetic and transformed into a strikingly natural movement.
At Johns Hopkins University, a group led by Michael McLoughlin is developing a mind-controlled device called the Modular Prosthetic Limb. It doesn’t require a brain implant, either. When the ability to control a limb is lost—whether to amputation, spinal-cord injury or another cause—the brain remembers how to use it. “We’re trying to establish a pathway to take that signal from the brain to the prosthetic,” McLoughlin says. They’re investigating non-invasive ways to tap into it, through electrodes or sensors implanted into the muscles or nerve bundles.
The Bionic Man’s ankles were designed by Hugh Herr, who’s been called a bionic man himself. In 1982, while attempting to summit Mount Washington in New Hampshire, Herr—then 17—was trapped in a vicious storm and lost both legs below the knees to frostbite. Now head of the biomechatronics research group at the MIT Media Lab in Boston, he designs advanced prostheses including the BiOM, the only commercially available bionic ankle device (Herr also wears them).
Manufactured by Media Lab spinoff company iWalk, of which Herr is founder and CEO, the BiOM propels users forward as they walk, similarly to how muscles behave in a biological leg. With his pant legs rolled down, it’s virtually impossible to tell that Herr is wearing prosthetics. “Would I wish my legs back? Absolutely not,” he says in the documentary, shortly after his nimble ascent of a rock-climbing wall. (Herr has three different sets of prosthetics just for climbing, he says.) “I think normal bodies are boring.”
As the pace of discovery moves ever faster, some scientists are already attempting to build souped-up versions of us, with abilities we’d never see in nature. Michael McAlpine, a professor of mechanical and aerospace engineering at Princeton University, recently used an off-the-shelf 3D printer to make an ear. That was remarkable enough, but this wasn’t a normal ear. Two very different “inks” were fed into the printer: cells (calf cells, in this case) and a paste of silver nanoparticles that formed a coiled antenna inside the cartilage. Beyond the implications for those with hearing difficulties, his antenna can receive electromagnetic signals and pipe them directly to the brain, which could bestow a sort of superpowered hearing, an “electronic sixth sense,” McAlpine says, tuning into what bats or dolphins hear.
For thousands of years, humans have worn prosthetics; ancient Egyptian mummies have been found with wood-and-leather toes. Now that we’re able to tinker with the very building blocks of our bodies, we can alter ourselves in ways that once would have been unimaginable. As Rich Walker assembled the Bionic Man, plugging in its artificial organs and attaching pneumatic artificial muscle to prosthetic limbs, he pondered this. “How much of the human can you replace,” he wondered, “before it’s a different human?”
